PeakSonic M1 & M2 Bladder Scanner - Intelligent Accuracy in the Palm of Your Hand
Compact 3D handheld bladder scanner with AI-assisted modes and ±7% accuracy, designed for fast, non-invasive assessments in care homes, hospitals, community care, and clinical settings.
What Makes PeakSonic Bladder Scanners Different
Our bladder scanners combine advanced 3D ultrasound technology, an intuitive point-and-click workflow, and industry-leading ±7% accuracy to deliver fast, reliable bladder volume assessment across care homes and clinical settings.
Key Benefit
Advanced 3D Bladder Imaging for precise bladder volume assessment.
Key Benefit
Industry-Leading Scan Accuracy, outperforming the ±14% industry benchmark.
Key Benefit
Rapid Clinical Results, supporting faster clinical decisions.
Key Benefit
Simple Point-and-Click Operation for ease of use and rapid staff adoption.
Key Benefit
Non-Invasive Patient Assessment, reducing reliance on catheterisation.
Key Benefit
Bladder Wall Thickness Measurement (M5) for advanced clinical insight
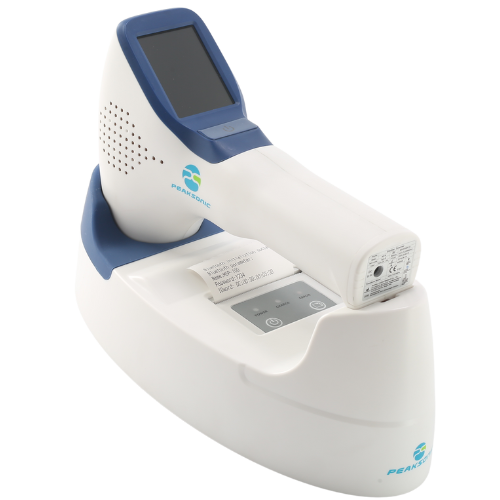
Accurate and Portable Bladder Scanning
Purpose-built with innovation and usability at its core, the PeakSonic M1 & M2 Bladder Scanners set a new standard for reliability, precision, and ease of use in urological assessments.
A key advantage is Intelligent Mode—an exclusive pre-scan function that helps clinicians locate the bladder with greater precision before measurement.
With this capability, the M1 & M2 Bladder Scanners deliver industry-leading accuracy of ±7%, significantly outperforming the ±14% benchmark commonly seen across comparable devices.
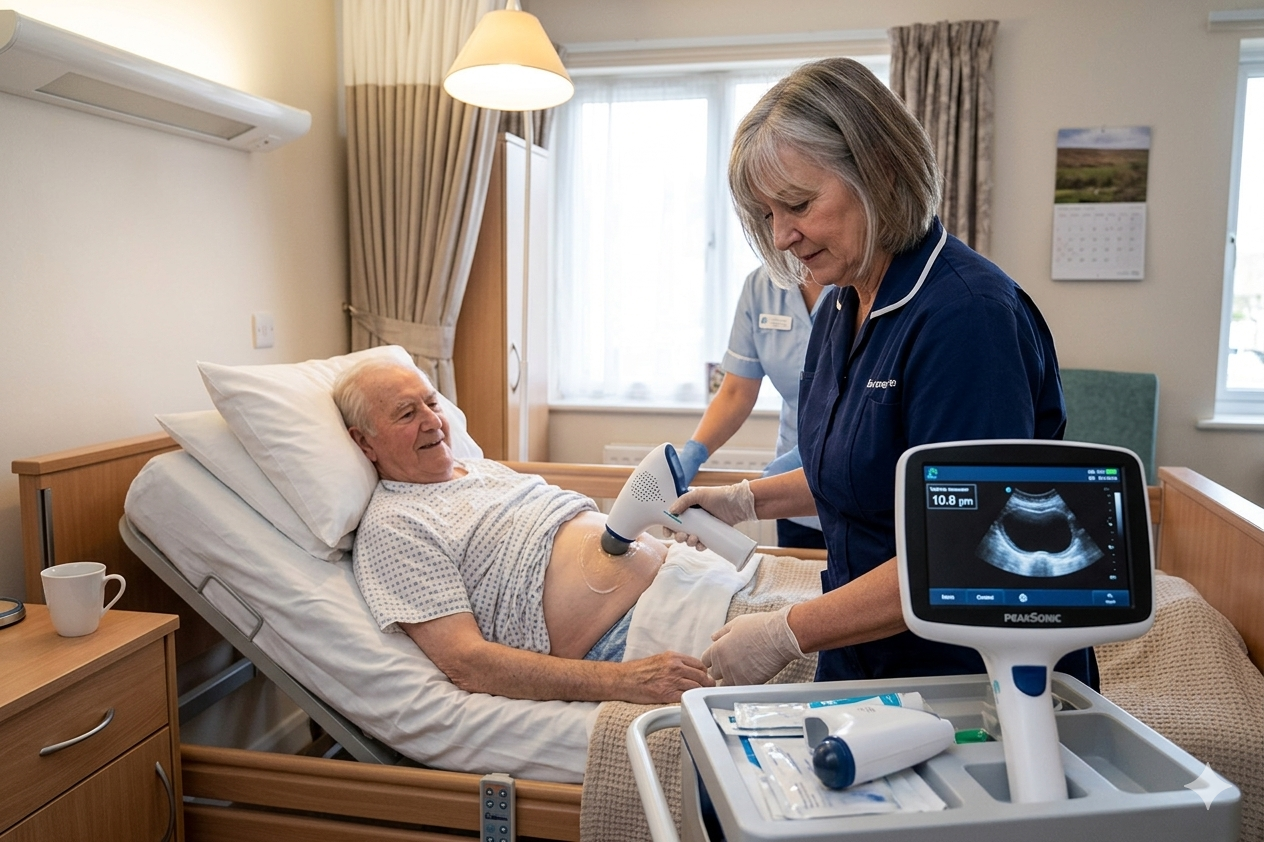
Simple, User-Friendly Bladder Scanning Operation
The M1 & M2 are designed for seamless integration into daily clinical routines, with an intuitive interface and minimal setup that allow trained healthcare professionals to perform scans quickly and confidently without disrupting workflows.
Portable and Lightweight Bladder Scanner Design
Lightweight and easy to move between rooms, the PeakSonic M1 & M2 are ideal for bedside assessments, multi-ward use, and community or mobile healthcare settings, with a compact footprint that suits facilities with limited space.
Clinical Accuracy, Compliance & Governance Confidence
The scanners are CE/UKCA marked and compliant with UK medical device requirements, making it suitable for regulated healthcare environments including the NHS, with supporting documentation available for governance and procurement.
Who Is It For?
The PeakSonic M1/M2 are designed for healthcare professionals who need fast, non-invasive bladder volume and post-void residual (PVR) assessment at the point of care. Manufacturer materials position it for hospitals, emergency care, obstetrics, gynecology, radiotherapy, nursing homes, rehabilitation centres, and elderly care settings.

PeakSonic M1 and M2 Bladder Scanner Specifications


Clinical Use Cases for M1 and M2 Bladder Scanners
The scanner can be applied in Surgery, Emergency, ICU, Obstetrics, Gynecology, radiotherapy and nursing homes.
It can provide the basis for clinical catheterization, evaluates the post-void residual after patient urination, and helps to make an auxiliary diagnosis on bladder and kidney diseases.
It is also suitable for patients with lower body disability or loss of auto-urination function to confirm the proper timing of urination.
Bladder Scanning Modes and Operation

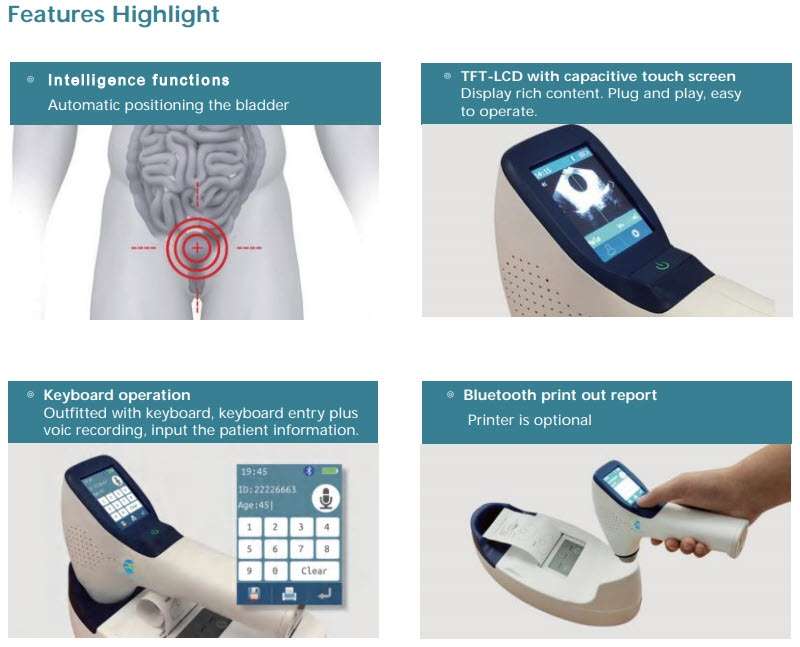
Key Features of PeakSonic M1 and M2 Bladder Scanners
M1 & M2 Models
M1-W & M2-W Models
Highlighted Features of M1 and M2 Models
M1 and M2 vs Traditional Bladder Scanners
FeatureS | M1 & M2 Scanner | Traditional Catheterization |
|---|---|---|
Non-Invasive | YES | NO |
Scan Time | Under 5 seconds | 10–15 minutes |
CAUTI Risk | NONE | HIGH |
Portability | Handheld | Not Applicable |
Data Management | 10,000+ cases | MANUAL |
Accuracy Rate | ±7% | ±14% |
Comes with a Dedicated Portable Carry Bag
The PeakSonic M1 & M2 are supplied with a dedicated portable carry bag, providing safe storage and easy transport between rooms, wards, or community settings while helping protect the device during everyday clinical use.



